
Breaking News
 Fury as Trump gets $1.8 billion taxpayer-funded payout from his own government:
Fury as Trump gets $1.8 billion taxpayer-funded payout from his own government:
 Kyle Rittenhouse gets huge blowback from MAGA after supporting anti-Trump candidate...
Kyle Rittenhouse gets huge blowback from MAGA after supporting anti-Trump candidate...
 Aaron Rodgers is back! Quarterback officially signs mega-money deal with the Pittsburgh Steelers...
Aaron Rodgers is back! Quarterback officially signs mega-money deal with the Pittsburgh Steelers...
 Switzerland To Vote On Capping Population At 10 Million
Switzerland To Vote On Capping Population At 10 Million
Top Tech News
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
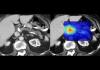 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
New ultrathin semiconductor materials exceed some of silicon's capabilities

In this greatly enlarged cross-section of an experimental chip, the bands of black and white reveal alternating layers of hafnium diselenide – an ultrathin semiconductor material – and the hafnium dioxide insulator. The cross-section matches an overlaid color schematic on the right. (Image credit: Michal Mleczko)
The new materials can also be shrunk to functional circuits just three atoms thick and they require less energy than silicon circuits. Although still experimental, the researchers said the materials could be a step toward the kinds of thinner, more energy-efficient chips demanded by devices of the future.
Science Advances – HfSe2 and ZrSe2: Two-dimensional semiconductors with native high-κ oxides
Silicon's strengths
Silicon has several qualities that have led it to become the bedrock of electronics, Pop explained. One is that it is blessed with a very good "native" insulator, silicon dioxide or, in plain English, silicon rust. Exposing silicon to oxygen during manufacturing gives chip-makers an easy way to isolate their circuitry. Other semiconductors do not "rust" into good insulators when exposed to oxygen, so they must be layered with additional insulators, a step that introduces engineering challenges. Both of the diselenides the Stanford group tested formed this elusive, yet high-quality insulating rust layer when exposed to oxygen.

 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone

