
Breaking News
 Justice Department Launches Criminal Investigation Into Funding E. Jean Carroll Received...
Justice Department Launches Criminal Investigation Into Funding E. Jean Carroll Received...
 They're Deliberately Destroying Men's Health. Here's the Antidote w/ Andrew Swedeger
They're Deliberately Destroying Men's Health. Here's the Antidote w/ Andrew Swedeger
 Freed Gaza Flotilla Activists Allege Israeli Abuse Including Rape; Israel Denies Claims
Freed Gaza Flotilla Activists Allege Israeli Abuse Including Rape; Israel Denies Claims
 'Feeding Our Future' Fraud Mastermind, Aimee Bock, Sentenced to 41 Years in Prison
'Feeding Our Future' Fraud Mastermind, Aimee Bock, Sentenced to 41 Years in Prison
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
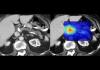 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
MIT tech could let desalination plants use their own brine waste

Developed by a research team at MIT, the proprietary system incorporates what are described as "well-known and standard chemical processes." These include a nanofiltration process to initially remove unwanted compounds from the brine, followed by one or more stages of electrodialysis. As a result, the brine is converted into useful chemicals such as sodium hydroxide.
More commonly known as caustic soda, sodium hydroxide is often utilized to change the acidity of seawater entering desalination plants, which in turn helps to prevent fouling of the filtration membranes that are used to remove the salt. Ordinarily, plant operators have to buy the chemical. With the MIT system, though, they would be able to produce even more than they need, on-site. The excess could then be sold for use in other applications.
Other chemicals that could be produced from the brine include hydrochloric acid, sales of which would likewise be a source of revenue. And as an added bonus, the scientists suggest that plants could save money by no longer needing to pump their brine out into the ocean.



