
Breaking News
 Is 'Project Freedom' Just Another Trump Scam?
Is 'Project Freedom' Just Another Trump Scam?
 THEY LIED About the Water - THE WELLS ARE GOING DRY GLOBALLY
THEY LIED About the Water - THE WELLS ARE GOING DRY GLOBALLY
 After Attack of Cargo Vessel, Trump Directs US to Escort Foreign Ships Through Hormuz
After Attack of Cargo Vessel, Trump Directs US to Escort Foreign Ships Through Hormuz
 RED ALERT: "I Think That You're Gonna See Billions Dead At This Rate!"
RED ALERT: "I Think That You're Gonna See Billions Dead At This Rate!"
Top Tech News
 Robot Dives 1.5 Miles, Maps French Shipwreck With 86,000 Images And Recovers Artifacts
Robot Dives 1.5 Miles, Maps French Shipwreck With 86,000 Images And Recovers Artifacts
 Brain-inspired chip could reduce AI energy use by 70%
Brain-inspired chip could reduce AI energy use by 70%
 "This is the first synthetic species," microbiologist J. Craig Venter told 60 Minutes'
"This is the first synthetic species," microbiologist J. Craig Venter told 60 Minutes'
 Humanoid robots are hitting the factories at an increasing pace
Humanoid robots are hitting the factories at an increasing pace
 Microsoft's $400 Billion Mistake Is Now a $200 Phone With Zero Tracking
Microsoft's $400 Billion Mistake Is Now a $200 Phone With Zero Tracking
 Turn Sand to Stone With Vinegar. Stronger Than Steel. Hidden Since 1627
Turn Sand to Stone With Vinegar. Stronger Than Steel. Hidden Since 1627
 This is a bioprinter printing with living human cells in real time
This is a bioprinter printing with living human cells in real time
 The remarkable initiative is called The Uncensored Library,...
The remarkable initiative is called The Uncensored Library,...
 Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Lithium-ion battery boost could come from "caging" silicon in graphene
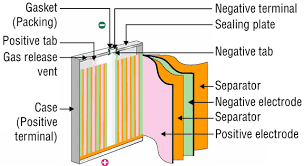
The researchers managed to remove two long-standing barriers to these improvements by putting silicon particles in graphene "cages."
To improve capacity in recent years batteries have begun to use silicon anodes, which have more capacity than the graphite conventionally used. But silicon particles also swell so much during charging that they're prone to cracking or shattering and they can also react with the battery electrolyte, forming a coating that reduces performance.
The solution from the team at Stanford and the Department of Energy's SLAC National Accelerator Laboratory is to encase each silicon particle in a "custom-fit cage" of graphene. At only one-atom thick, graphene is the thinnest, strongest form of carbon and also conducts electricity well.
The carbon cages would allow the silicon to expand and even break apart, but keep the pieces together so that they can continue to function. The graphene barrier would also block the destructive chemical reactions with the electrolyte from occurring.

 CANCER HAS BEEN CURED
CANCER HAS BEEN CURED

