
Breaking News
 The U.S. Empire's Motivations in the Ukraine War
The U.S. Empire's Motivations in the Ukraine War
 Polish Prime Minister Tusk Says NATO Alliance Faces 'Disintegration' Amid U.S. Troop Reducti
Polish Prime Minister Tusk Says NATO Alliance Faces 'Disintegration' Amid U.S. Troop Reducti
 Trump Will Make Europe (Especially Germany) Grateful Again
Trump Will Make Europe (Especially Germany) Grateful Again
Top Tech News
 Robot Dives 1.5 Miles, Maps French Shipwreck With 86,000 Images And Recovers Artifacts
Robot Dives 1.5 Miles, Maps French Shipwreck With 86,000 Images And Recovers Artifacts
 Brain-inspired chip could reduce AI energy use by 70%
Brain-inspired chip could reduce AI energy use by 70%
 "This is the first synthetic species," microbiologist J. Craig Venter told 60 Minutes'
"This is the first synthetic species," microbiologist J. Craig Venter told 60 Minutes'
 Humanoid robots are hitting the factories at an increasing pace
Humanoid robots are hitting the factories at an increasing pace
 Microsoft's $400 Billion Mistake Is Now a $200 Phone With Zero Tracking
Microsoft's $400 Billion Mistake Is Now a $200 Phone With Zero Tracking
 Turn Sand to Stone With Vinegar. Stronger Than Steel. Hidden Since 1627
Turn Sand to Stone With Vinegar. Stronger Than Steel. Hidden Since 1627
 This is a bioprinter printing with living human cells in real time
This is a bioprinter printing with living human cells in real time
 The remarkable initiative is called The Uncensored Library,...
The remarkable initiative is called The Uncensored Library,...
 Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
A Novel Aluminum–Graphite Dual-Ion Battery
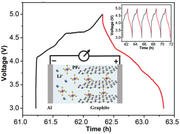
A packaged aluminum–graphite battery is estimated to deliver an energy density of ≈150 Wh kg−1 at a power density of ≈1200 W kg−1, which is ≈50% higher than most commercial lithium ion batteries.
Lithium ion batteries based on cation intercalation have been powering the increasingly mobile society for decades.[1] In a conventional lithium ion battery, the intercalation of lithium ions in both cathode (i.e., LiCoO2, LiFePO4) and anode (i.e., graphite, silicon) materials have been thoroughly studied, while the utilization of the anions in the electrolyte has drawn much less attention.[2] In fact, the phenomenon of anion intercalate into graphite by chemical or electrochemical means was discovered and proposed as a possible positive electrode for batteries by Rüdorff and Hofmann in 1938.[3] However, the anion intercalation was achieved by using high concentration acid solution as electrolyte, this brought serious safety issue that hindered its application.[4] In the 1990s, soon after the commercial application of lithium ion battery, Carlin et al. reported dual graphite intercalating molten electrolyte batteries that realized the application of anion intercalated graphite as positive electrode in batteries by using room temperature ionic liquids as electrolyte.[5] In the following decades, continuous progresses have been made in anion intercalated graphite based dual carbon batteries, such as investigation of anion intercalation in non-aqueous electrolyte, in situ characterization of the staged anion intercalation process, and systematic study of the intercalation of different anions into graphite.[6]

 Contrived Wars and No Peace
Contrived Wars and No Peace CANCER HAS BEEN CURED
CANCER HAS BEEN CURED

