
Breaking News
 Mercantilism: China and Beyond
Mercantilism: China and Beyond
 The Magician, the Martyr, and the 33% Solution: Why Every Trump Poll Slump Requires A Fake...
The Magician, the Martyr, and the 33% Solution: Why Every Trump Poll Slump Requires A Fake...
 'Bitcoin Isn't Going Anywhere': Trump Officials Discuss DOJ, FBI Refocus on Crypto Crime
'Bitcoin Isn't Going Anywhere': Trump Officials Discuss DOJ, FBI Refocus on Crypto Crime
 The Praxian Genocidal Kill Chain – Part 1
The Praxian Genocidal Kill Chain – Part 1
Top Tech News
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
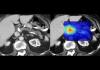 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone
 Wow. Researchers just built an AI that can control your body...
Wow. Researchers just built an AI that can control your body...
 Google Chrome silently installs a 4 GB AI model on your device without consent
Google Chrome silently installs a 4 GB AI model on your device without consent
 The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
The $5 Battery That Never Dies - Edison Buried This 100 Years Ago
 That is not a real fish. IT'S A ROBOT.
That is not a real fish. IT'S A ROBOT.
 Scientists Unveil Hemp Alternative to Plastic That Can Withstand Boiling Water...
Scientists Unveil Hemp Alternative to Plastic That Can Withstand Boiling Water...
Nanomembrane based tech can replace Haber-bosch process that feeds world

It operates under conditions of high temperature 400–500 °C and pressure 200–250 bar, and its production has a huge carbon footprint. The H2 precursor, usually obtained by steam reforming of methane, also has a very large carbon footprint. Notably, the entire energy required to prepare the reagents and to operate the Haber-Bosch process amounts to 1–3% of the global energy supply. In stark contrast, in the natural world, plants and bacteria have been producing NH3 from N2 and solvated protons under ambient conditions, enabled by the FeMo cofactor of the metalloenzyme nitrogenase (N2 + 6H+ 6e-→2NH3). Inspired by this biological nitrogen fixation process, intensive efforts have been devoted to finding ways to mimic the process under similarly mild conditions.
The Haber-Bosch process uses a catalyst or container made of iron or ruthenium with an inside temperature of over 800?F (426?C) and a pressure of around 200 atmospheres to force nitrogen and hydrogen together. The elements then move out of the catalyst and into industrial reactors where the elements are eventually converted into fluid ammonia (Rae-Dupree, 2011). The fluid ammonia is then used to create fertilizers.

 A Robot Economy: Who Gets Rich, Who Gets Left Behind
A Robot Economy: Who Gets Rich, Who Gets Left Behind

