
Breaking News
 For the first time in history, the majority of U.S. Stock Trading occurs off exchange
For the first time in history, the majority of U.S. Stock Trading occurs off exchange
 Catherine Herridge just exposed Directed Energy Weapons (DEWs)!
Catherine Herridge just exposed Directed Energy Weapons (DEWs)!
 Today, one of the top cryptocurrencies, Zcash, crashed over 40% overnight
Today, one of the top cryptocurrencies, Zcash, crashed over 40% overnight
 Gold just overtook US Treasuries as the world's top reserve asset.
Gold just overtook US Treasuries as the world's top reserve asset.
Top Tech News
 Every hard drive you own will die.
Every hard drive you own will die.
 Flying car industry turns to solid-state batteries for commercial takeoff
Flying car industry turns to solid-state batteries for commercial takeoff
 Thumbnail-sized thrusters could take CubeSats to Mars
Thumbnail-sized thrusters could take CubeSats to Mars
 Tesla Discovered How to Destroy Disease With Sound. Then They Buried It.
Tesla Discovered How to Destroy Disease With Sound. Then They Buried It.
 World's longest-range airliner takes to the skies
World's longest-range airliner takes to the skies
 Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
Microwave Water Splitting for Breakthroughs for Making Hydrogen, Oxygen and Fast Battery
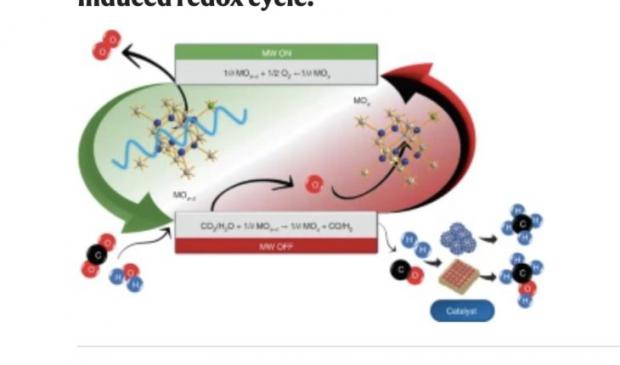
Researchers from the Polytechnic University of Valencia and the Spanish National Research Council (CSIC) report contactless H2 production via water electrolysis mediated by the microwave-triggered redox activation of solid-state ionic materials at low temperatures (less than 250 °C).
This will simplify and significantly cheapen the process for hydrogen production.
Water was reduced via reaction with non-equilibrium gadolinium-doped CeO2 that was previously in situ electrochemically deoxygenated by the sole application of microwaves. The microwave-driven reduction was identified by an instantaneous electrical conductivity rise and O2 release. This process was cyclable, whereas H2 yield and energy efficiency were material- and power-dependent. Deoxygenation of low-energy molecules (H2O or CO2) led to the formation of energy carriers and enabled CH4 production when integrated with a Sabatier reactor. This method could be extended to other reactions such as intensified hydrocarbons synthesis or oxidation.

 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...

