
Breaking News
 Molecular Hydrogen -- Is It the Best Antioxidant You Can Take?
Molecular Hydrogen -- Is It the Best Antioxidant You Can Take?
 Houthis Declare "Total Ban" On Israeli Ships As Dual Chokepoint Crisis Stokes Supply Chain
Houthis Declare "Total Ban" On Israeli Ships As Dual Chokepoint Crisis Stokes Supply Chain
 Nithya Raman Flips the LA Mayoral Race with a Stunning 43,000 Vote Swing...
Nithya Raman Flips the LA Mayoral Race with a Stunning 43,000 Vote Swing...
 BREAKING: Ceasefire COLLAPSES as Iran, Israel trade strikes
BREAKING: Ceasefire COLLAPSES as Iran, Israel trade strikes
Top Tech News
 World's longest-range airliner takes to the skies
World's longest-range airliner takes to the skies
 Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
39-Year-Old Becomes First US Patient to Receive Innovative Artificial Heart Prosthetic
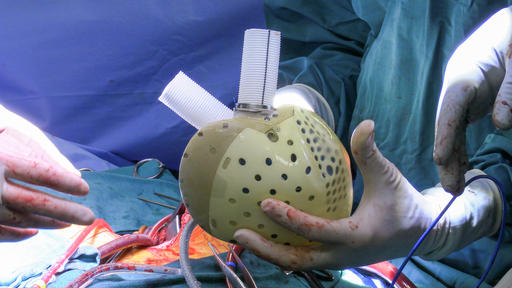
The artificial heart was developed by a French company, CARMAT and has been approved for use and sale in Europe.
Last year, the company received U.S. FDA approval to begin studies and enroll 10 patients with end-stage biventricular heart failure—people who are suffering on the waiting list for a heart donor—and offer a life-saving bridge before transplant.
"We are encouraged that our patient is doing so well after the procedure Monday," said Dr. Carmelo Milano, a transplant surgeon and the principal investigator of the device study at Duke. "As we evaluate this device, we are both excited and hopeful that patients who otherwise have few to no options could have a lifeline."
The North Carolina patient, Matthew Moore, is just 39-years-old and was referred to Duke in June after a sudden, unexpected diagnosis of heart failure. Moore and his wife, Rachel, recently adopted their two-year-old foster son, Marshall, and arrived at Duke expecting only to undergo heart bypass surgery.

 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater

