
Breaking News
 Woman flies to Seattle to show how all the businesses have left their downtown...
Woman flies to Seattle to show how all the businesses have left their downtown...
 James Freeman ILLEGAL ARREST DROPPED & HUGE LAWSUIT
James Freeman ILLEGAL ARREST DROPPED & HUGE LAWSUIT
 Jamie Kennedy blasts LA mayoral election swing: 'Literal crime scene'
Jamie Kennedy blasts LA mayoral election swing: 'Literal crime scene'
 Here we go, the Los Angeles Times is admitting that yes, tens of thousands of mail in ballots...
Here we go, the Los Angeles Times is admitting that yes, tens of thousands of mail in ballots...
Top Tech News
 World's longest-range airliner takes to the skies
World's longest-range airliner takes to the skies
 Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Nanostructured electrode could boost lithium battery storage by 50%
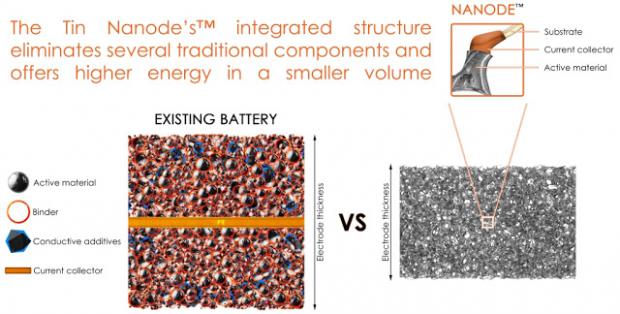
This allows the batteries to last longer between charges while also charging faster. These achievements are due to both the material structure and the use of tin as the active material. Tin is known to have much higher energy density than the current graphite technology, but until now its commercial success has been limited due to its tendency to swell during charging, causing stress in the electrode material and leading to a rapid loss in energy. Current commercial lithium ion batteries employ a foil/particle system as the electrode structure. The capability of such electrodes to deal with volume expansion of high energy materials is limited, because as the particles swell, the electrode expands.
The Tin Nanode's™ integrated electrode structure contributes to the relaxation of stress associated with electrode materials undergoing high volume expansion. This is possible because thin films of active material are spread over a 3D and porous network of fibres, rather than stacking particles on a flat copper foil.

 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater

