
Breaking News
 U.S. sends aircraft carrier Nimitz to the Caribbean as pressure mounts on Cuba
U.S. sends aircraft carrier Nimitz to the Caribbean as pressure mounts on Cuba
 'Why the f**k should I support this guy?' Trump's revenge against Thune…
'Why the f**k should I support this guy?' Trump's revenge against Thune…
 The Grocery Store Is Becoming a Surveillance Center
The Grocery Store Is Becoming a Surveillance Center
 Will Affordability Bankrupt President Trump?
Will Affordability Bankrupt President Trump?
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
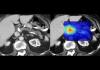 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Insulator Becomes Conducting Semiconductor And Could Make Superelastic Silicone Solar Panels
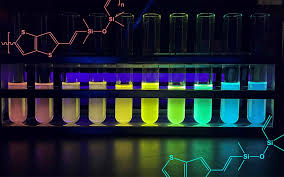
On a molecular level, silicones are made up of a backbone of alternating silicon and oxygen atoms (Si—O—Si) with organic (carbon-based) groups attached to the silicon. Various 3D formations of polymer chains arise as they connect to one another, known as cross-linking, which alter the material's physical properties like strength or solubility.
This could lead to super-elastic silicone PV cells(/panels) and super-elastic thus super-durable, thick and highly energy dense lithium-ion battery anodes.
While studying different cross-linking structures in silicone, the research team stumbled upon the potential for electrical conductivity in a copolymer, which is a polymer chain containing two different types of repeating units—cage-structured and then linear silicones in this case.
The possibility for conductivity arises from the way electrons can move across Si—O—Si bonds with overlapping orbitals. Semiconductors have two main states: the ground state, which doesn't conduct electricity, and a conducting state, which does. The conducting state, also known as an excited state, occurs when some electrons jump up to the next electron orbital, which is connected across the material like a metal.
Typically, Si—O—Si bond angles don't allow for that connection. At 110°, they are a long way from a 180° straight line. But in the silicone copolymer the team discovered, these bonds started out at 140° in the ground state—and they stretch to 150° in the excited state. This was enough to create a highway for electrical charge to flow.
σ–σ* conjugation Across Si?O?Si Bonds. Macromol.
"This allows an unexpected interaction between electrons across multiple bonds including Si—O—Si bonds in these copolymers," Laine said. "The longer the chain length, the easier it is for electrons to travel longer distances, reducing the energy needed to absorb light and then emit it at lower energies."
The semiconducting properties of the silicone copolymers also enable its spectrum of colors. Electrons jump between the ground and excited states by absorbing and emitting photons, or particles of light. The light emission depends on the length of the copolymer chain, which Laine's team can control. Longer chain lengths mean smaller jumps and lower energy photons, giving the silicone a red tint. Shorter chains require bigger jumps from the electrons, so they emit higher energy light toward the blue end of the spectrum.



