
Breaking News
 If housing prices had simply followed income growth since 1970
If housing prices had simply followed income growth since 1970
 According to INSEE data, France is now channeling 57.2% of its GDP through government spending
According to INSEE data, France is now channeling 57.2% of its GDP through government spending
 Power is moving eastwards and westerners are talking about how great it is...
Power is moving eastwards and westerners are talking about how great it is...
 Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Top Tech News
 Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
 Interceptor-Drone Arms-Race Emerges
Interceptor-Drone Arms-Race Emerges
 A startup called Inversion has introduced Arc, a space-based vehicle...
A startup called Inversion has introduced Arc, a space-based vehicle...
 Mining companies are using cosmic rays to find critical minerals
Mining companies are using cosmic rays to find critical minerals
 They regrew a severed nerve - by shortening a bone.
They regrew a severed nerve - by shortening a bone.
 New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
 Russian scientists 'are developing the world's first drug to delay ageing' months after
Russian scientists 'are developing the world's first drug to delay ageing' months after
 Sam Altman's World ID Expands Biometric Identity Checks
Sam Altman's World ID Expands Biometric Identity Checks
 China Tests Directed Energy Beam That Recharges Drones Mid-Flight
China Tests Directed Energy Beam That Recharges Drones Mid-Flight
 Jurassic Park might arrive sooner than expected, just with Dinobots.
Jurassic Park might arrive sooner than expected, just with Dinobots.
Parsley and vanilla beans are being used as cellular scaffolding to grow stem cell tissues
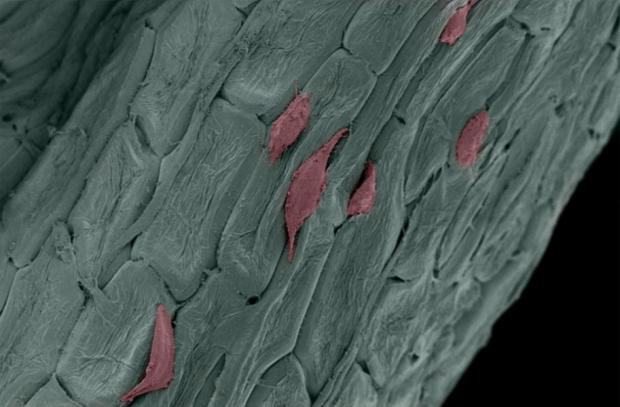
Researchers at the University of Washington-Madison were able to grow skin, brain, bone marrow and blood vessels on plants using a highly-specialized, natural scaffolding from plants like parsley. The team observed that certain plant species possess strength, rigidity and porosity as well as low mass and surface area. These characteristics make for a structurally-efficient scaffold. Plants have really high surface area to volume ratio, while their porous structure facilitates fluid transport, a study author said. The researchers also found that the 3D printed stem cell scaffold helped support, feed and organize the cells.
The researchers have collaborated with the Olbrich Botanical Gardens to identify plant species that show scaffolding potential, which in turn could be turned into structures for biomedical purposes. John Wirth, Olbrich's conservatory curator, said the idea was a good way to use the living plant material to develop human tissue. Parsley, orchid, and vanilla were among the plant species chosen for the study. Bamboo, wasabi, and elephant ear plant were also among the plants were cellulose was derived. "Nature provides us with a tremendous reservoir of structures in plants. You can pick the structure you want," said Gianluca Fontana, the lead author of the new study and a UW-Madison postdoctoral fellow.
"Plants have a huge capacity to grow cell populations. They can deliver fluids very efficiently to their leaves … At the microscale, they're very well organized," said Bill Murphy, co-director of the UW-Madison Stem Cell and Regenerative Medicine Center.
"The vast diversity in the plant kingdom provides virtually any size and shape of interest. It really seemed obvious. Plants are extraordinarily good at cultivating new tissues and organs, and there are thousands of different plant species readily available. They represent a tremendous feedstock of new materials for tissue engineering applications," Murphy added.
Study details: Cellulose and 3D scaffolding techniques
The researchers decellularized the plant materials leaving only cellulose, the basic components of a plant's cell walls. The team then added peptides to serve as biological fasteners since human cells have no affinity to cellulose. Advanced technologies such as 3D printing and injection molding were used to create the three-dimensional scaffolds.
It was found that eliminating all the other cells that make up the plant and retaining only the cellulose husks encouraged human stem cells such as fibroblasts to attach to the scaffold and develop miniature structures. Fibroblasts are common connective tissue cells that result from stem cell cultivation.
Stem cells seeded into the scaffold also appeared to align themselves along its structure. This mechanism indicates a potential to use the materials in order to regulate the structure and alignment of developing human tissues, which may prove crucial for nerve and muscle tissues that need alignment and patterning. "Stem cells are sensitive to topography. It influences how cells grow and how well they grow," lead author Gianluca Fontana said.



