
Breaking News
 If housing prices had simply followed income growth since 1970
If housing prices had simply followed income growth since 1970
 According to INSEE data, France is now channeling 57.2% of its GDP through government spending
According to INSEE data, France is now channeling 57.2% of its GDP through government spending
 Power is moving eastwards and westerners are talking about how great it is...
Power is moving eastwards and westerners are talking about how great it is...
 Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Top Tech News
 Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
 Interceptor-Drone Arms-Race Emerges
Interceptor-Drone Arms-Race Emerges
 A startup called Inversion has introduced Arc, a space-based vehicle...
A startup called Inversion has introduced Arc, a space-based vehicle...
 Mining companies are using cosmic rays to find critical minerals
Mining companies are using cosmic rays to find critical minerals
 They regrew a severed nerve - by shortening a bone.
They regrew a severed nerve - by shortening a bone.
 New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
 Russian scientists 'are developing the world's first drug to delay ageing' months after
Russian scientists 'are developing the world's first drug to delay ageing' months after
 Sam Altman's World ID Expands Biometric Identity Checks
Sam Altman's World ID Expands Biometric Identity Checks
 China Tests Directed Energy Beam That Recharges Drones Mid-Flight
China Tests Directed Energy Beam That Recharges Drones Mid-Flight
 Jurassic Park might arrive sooner than expected, just with Dinobots.
Jurassic Park might arrive sooner than expected, just with Dinobots.
A Brilliant New Cancer Treatment That Re-Engineers Human Cells Just Got Approved
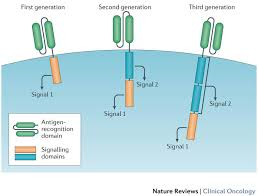
The US Food and Drug Administration (FDA) just approved a cutting-edge cancer therapy.
On Wednesday, the FDA approved Novartis's Kymriah, also known as tisagenlecleucel, a treatment for pediatric acute lymphoblastic lymphoblastic leukemia.
"I think this is most exciting thing I've seen in my lifetime," Dr. Tim Cripe, an oncologist who was part of the FDA advisory committee panel that voted in favour of approving the drug in July.
The highly personalised treatment is called CAR T-cell therapy. It's a type of cancer immunotherapy — or a therapy that harnesses the body's immune system to take on cancer cells.
"We're entering a new frontier in medical innovation with the ability to reprogram a patient's own cells to attack a deadly cancer," FDA commissioner Scott Gottlieb said in a statement.



