
Breaking News
 If housing prices had simply followed income growth since 1970
If housing prices had simply followed income growth since 1970
 According to INSEE data, France is now channeling 57.2% of its GDP through government spending
According to INSEE data, France is now channeling 57.2% of its GDP through government spending
 Power is moving eastwards and westerners are talking about how great it is...
Power is moving eastwards and westerners are talking about how great it is...
 Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Agricultural commodities are now breaking out decisively from nearly 20-year resistance.
Top Tech News
 Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
Researcher wins 1 bitcoin bounty for 'largest quantum attack' on underlying tech
 Interceptor-Drone Arms-Race Emerges
Interceptor-Drone Arms-Race Emerges
 A startup called Inversion has introduced Arc, a space-based vehicle...
A startup called Inversion has introduced Arc, a space-based vehicle...
 Mining companies are using cosmic rays to find critical minerals
Mining companies are using cosmic rays to find critical minerals
 They regrew a severed nerve - by shortening a bone.
They regrew a severed nerve - by shortening a bone.
 New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
New Robot Ants Work Like Real Insects To Build And Dismantle On Their Own
 Russian scientists 'are developing the world's first drug to delay ageing' months after
Russian scientists 'are developing the world's first drug to delay ageing' months after
 Sam Altman's World ID Expands Biometric Identity Checks
Sam Altman's World ID Expands Biometric Identity Checks
 China Tests Directed Energy Beam That Recharges Drones Mid-Flight
China Tests Directed Energy Beam That Recharges Drones Mid-Flight
 Jurassic Park might arrive sooner than expected, just with Dinobots.
Jurassic Park might arrive sooner than expected, just with Dinobots.
Tissue engineering, replacement organs and regenerative medicine are getting friendlier regulations
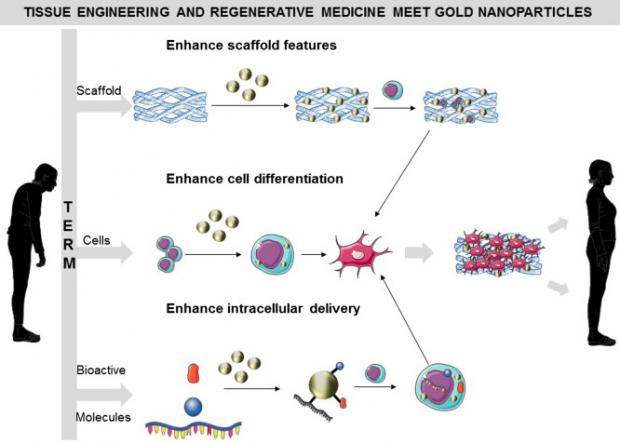
The FDA intends to promote the "least burdensome" rules for companies big and small that are seeking to develop new therapies, "while ensuring patient safety."
"Our policy will allow product manufacturers that time to engage with the FDA to determine if they need to submit a marketing authorization application and, if so, seek guidance on how to submit their application to the FDA for approval," Gottlieb said.
The new rules are in keeping with provisions from the 21st Century Cures Act, passed by Congress in December. That legislation earmarked $6.3 billion in funding, mostly for the U.S. National Institutes of Health, towards groundbreaking medical research.
Over the past few years scientists and physicians have developed tissue-engineered skin for transplant; bladders grown from a patient's own cells; and tissues grown to repair ailing hearts or failing knees.



