
Breaking News
 UN IPCC climate group reverses course on doomsday predictions...
UN IPCC climate group reverses course on doomsday predictions...
 False Flag Alert! U.S. Intelligence Claims Cuba Considering Drone Strike On Key West Florida...
False Flag Alert! U.S. Intelligence Claims Cuba Considering Drone Strike On Key West Florida...
 Exclusive -- Robert F. Kennedy Jr.: Biden Admin's 453 Pages of Food Guidelines...
Exclusive -- Robert F. Kennedy Jr.: Biden Admin's 453 Pages of Food Guidelines...
 Comedian replacing Stephen Colbert appears to take a swipe at his predecessor as he vows...
Comedian replacing Stephen Colbert appears to take a swipe at his predecessor as he vows...
Top Tech News
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
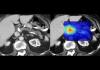 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
A multi-terrain robot from China is going viral, not because of raw speed or power...
New evidence strengthens link between telomere length, aging and cancer
Now a new study has found an intriguing piece of evidence supporting this hypothesis in genomes from several families that seem to be particularly prone to cancer.
In a way, our cells have a pre-determined number of divisions in their lifetime – around 50. That limit is dictated by our telomeres, small repeating segments of "junk" DNA that form caps on the ends of our chromosomes. These act like a buffer protecting the important DNA in the chromosomes from damage when a cell divides, but a little piece of the telomere is lost each time.
Eventually that damage adds up and the telomeres shorten to the point that the cell stops dividing. This contributes to the symptoms of aging that we're all too familiar with.
In theory, lengthening our telomeres or preventing them from shrinking should help slow the aging process, or even reverse it. Indeed, plenty of research is investigating this angle. But there's a nasty potential downside to doing so – cancer.
Cancer cells are effectively immortal, in the sense that they never stop dividing. It appears that having telomeres of a set length is an evolutionary defense mechanism to prevent that kind of runaway growth. And the new study has found more evidence supporting that hypothesis.
Researchers at the Rockefeller University and the Radboud University Medical Center studied the genomes of several Dutch families that appeared to be quite cancer-prone. Common among these patients were mutations in a gene called TINF2, which codes for a protein previously linked to telomere length.
So the team used CRISPR to engineer human cells with the same mutations, and found that they had much longer telomeres than usual. When the scientists checked the patients themselves, these little caps were also found to be particularly long.

 The World's Biggest Fusion Reactor Just Hit A Milestone
The World's Biggest Fusion Reactor Just Hit A Milestone

