
Breaking News
 For the first time in history, the majority of U.S. Stock Trading occurs off exchange
For the first time in history, the majority of U.S. Stock Trading occurs off exchange
 Catherine Herridge just exposed Directed Energy Weapons (DEWs)!
Catherine Herridge just exposed Directed Energy Weapons (DEWs)!
 Today, one of the top cryptocurrencies, Zcash, crashed over 40% overnight
Today, one of the top cryptocurrencies, Zcash, crashed over 40% overnight
 Gold just overtook US Treasuries as the world's top reserve asset.
Gold just overtook US Treasuries as the world's top reserve asset.
Top Tech News
 Every hard drive you own will die.
Every hard drive you own will die.
 Flying car industry turns to solid-state batteries for commercial takeoff
Flying car industry turns to solid-state batteries for commercial takeoff
 Thumbnail-sized thrusters could take CubeSats to Mars
Thumbnail-sized thrusters could take CubeSats to Mars
 Tesla Discovered How to Destroy Disease With Sound. Then They Buried It.
Tesla Discovered How to Destroy Disease With Sound. Then They Buried It.
 World's longest-range airliner takes to the skies
World's longest-range airliner takes to the skies
 Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
Batteries That Use Sodium Instead of Lithium Could Be Low-Cost Rival to Tesla's
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
Progress to Practical Room Temperature Superconductors
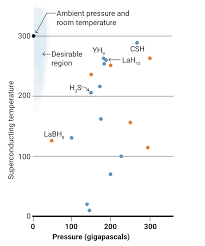
Theorists have predicted other hydride compounds which could work at lower pressures. There is race to find versions stable at ambient pressure and room temperature.
In 2004, Ashcroft suggested that adding other elements to hydrogen might add a "chemical precompression," stabilizing the hydrogen lattice at lower pressures. The race was on to make superconducting hydrides. In 2015, researchers including Mikhail Eremets, a physicist at the Max Planck Institute for Chemistry, reported in Nature that a mix of sulfur and hydrogen superconducted at 203 K when pressurized to 155 GPa. Over the next 3 years, Eremets and others boosted the Tc as high as 250 K in hydrides containing the heavy metal lanthanum. Then came Dias's CSH compound, reported late last year in Nature, which superconducts at 287 K—or 14°C, the temperature of a wine cellar—under 267 GPa of pressure, followed by an yttrium hydride that superconducts at nearly as warm a temperature, announced by multiple groups this year.

 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...

