
Breaking News
 How to Make Infinite FREE Chicken Feed
How to Make Infinite FREE Chicken Feed
 Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
Amish's 25 BANNED Epsom Salt Hacks from the 1950s That Doctors Now Say Were GENIUS
 The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
The Korean Bucket That Grows Unlimited Plant Steroids -- Doubles Every 48 Hours
 This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
This CHICKEN Lays 365 EGGS Per Year. Never Gets Sick. Why Did They Make It Disappear?
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
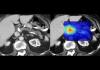 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
The FDA's War on America's Health

For most of my life, I've observed the FDA belligerently suppress natural treatments and any unorthodox therapy which threatens the medical monopoly while simultaneously railroading through a variety of unsafe and ineffective drugs regardless of how much public protest the agency meets. Consider this 2004 Senate testimony by the FDA scientist who got Vioxx banned that accurately described exactly what would come to pass with the COVID vaccines two decades later.
As such, I do not hold the FDA in a positive light, especially given that during COVID-19, I (like many others) spent hundreds of hours trying to get the agency to allow the limited use of off-patent therapeutics for COVID-19—all of which ultimately went nowhere due to the unjustifiable roadblocks the agency kept putting up.
Over the past year, especially since Trump's election, I've received many questions about FDA reform. To address the issue properly, I've carefully examined both sides.
In medicine, "sensitivity" refers to a test's ability to correctly identify those who have a condition (e.g., detecting an infection), while "specificity" measures how well the test avoids false positives (i.e., correctly identifying those who don't have the condition). The challenge is that improving one often reduces the other. For example, increasing the PCR cycle threshold in COVID tests made it more likely to detect infections (higher sensitivity), but also increased false positives (lower specificity). This trade-off leads to problems, like breast cancer screenings, where high sensitivity can result in false positives and unnecessary "treatments" for women who don't actually have cancer.
The FDA faces a similar challenge: it must prevent harmful foods and drugs from reaching the market while ensuring useful products aren't blocked. Though this seems straightforward, it's incredibly difficult, and the FDA has often failed at both, even with leadership dedicated to public health.
Crime Against the Food Law
In the late 1800s, food producers were selling adulterated products, and pharmaceutical companies peddled medicines with secret ingredients like opium and alcohol. Public outrage grew, especially after exposés like Upton Sinclair's The Jungle, which helped spark the 1906 Pure Food and Drug Act. This law gave the Bureau of Chemistry the power to ensure accurate labeling and prevent harmful additives in food.
The director of the Bureau of Chemistry (and thus the first head of the FDA), Harvey Wiley conducted tests on food additives, proving they made healthy volunteers sick. While the public and many scientists supported his findings, the food industry fought back with powerful lobbyists and legal tactics.



