
Breaking News
 The Judaism-Zionism Bifurcation: Tikkun Olam: Fixing The World, But For What, For Whom?
The Judaism-Zionism Bifurcation: Tikkun Olam: Fixing The World, But For What, For Whom?
 Aww... Look At The Cute Dancing-Robot Police-State Surveillance-Dog...
Aww... Look At The Cute Dancing-Robot Police-State Surveillance-Dog...
 "Working Better": Saylor Teases BTC Buy After Strategy Sells For First Time Since 2022
"Working Better": Saylor Teases BTC Buy After Strategy Sells For First Time Since 2022
Top Tech News
 Elon and SpaceX Have Made AI Training 10 Times Faster
Elon and SpaceX Have Made AI Training 10 Times Faster
 Oklo COO Says Nuclear Waste Could Power America For 150 Years
Oklo COO Says Nuclear Waste Could Power America For 150 Years
 SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
SpaceX Announces LARGEST Starship Mission Ever! They've never done this before!
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New catalyst rearranges carbon dioxide and water into ethanol fuel
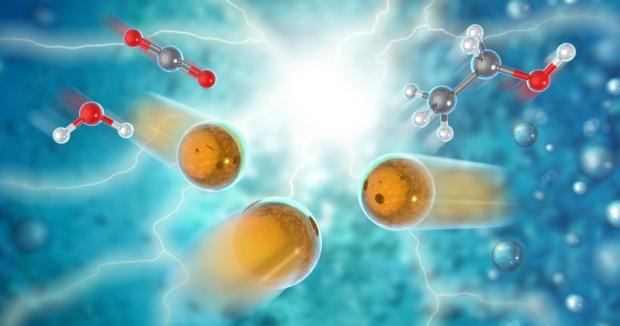
Researchers at the US Dept of Energy's Argonne National Laboratory, working with Northern Illinois University, have discovered a new catalyst that can convert carbon dioxide and water into ethanol with "very high energy efficiency, high selectivity for the desired final product and low cost."
The catalyst is made of atomically dispersed copper on a carbon-powder support, and acts as an electrocatalyst, sitting in a low voltage electric field as water and carbon dioxide are passed over it. The reaction breaks down these molecules, then selectively rearranges them into ethanol with an electrocatalytic selectivity, or "Faradaic efficiency", higher than 90%. The team says this is "much higher than any other reported process."
Once the ethanol is created, it can be used as a fuel additive, or as an intermediate product in the chemical, pharmaceutical and cosmetics industries. Using it as a fuel would be an example of a "circular carbon economy," in which CO2 recaptured from the atmosphere is effectively put back in as it's burned.
If the process is powered by renewable energy, which the researchers say it can be due to its low-temperature, low-pressure operation and easy responsiveness to intermittent power, then great; all you're losing is fresh water, which is its own issue.

 The Trump Hustle
The Trump Hustle


