
Breaking News
 Thomas Massie delivers concession speech after losing Kentucky primary to Ed Gallrein (PERFECT!)
Thomas Massie delivers concession speech after losing Kentucky primary to Ed Gallrein (PERFECT!)
 Apocalypse Watch E236: The UFO Slow Roll: Disclosure or Distraction?
Apocalypse Watch E236: The UFO Slow Roll: Disclosure or Distraction?
 BYD's New Blade Battery Is Brilliant, But Good Luck Taking It Apart
BYD's New Blade Battery Is Brilliant, But Good Luck Taking It Apart
Top Tech News
 Cars Are Fast Becoming Dystopian Prison Pods...
Cars Are Fast Becoming Dystopian Prison Pods...
 Our Emergency Water Plan Wasn't Good Enough - So We Built This
Our Emergency Water Plan Wasn't Good Enough - So We Built This
 Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
Sodium Ion Batteries Can Reach 100 Gigawatt Per Hour Per Year Scale in 2027
 Juiced Bikes proves capable electric motorcycles don't have to cost a lot
Juiced Bikes proves capable electric motorcycles don't have to cost a lot
 Headlight projectors turn your car into a drive-in theater
Headlight projectors turn your car into a drive-in theater
 US To Develop Small Modular Nuclear Reactors For Commercial Shipping
US To Develop Small Modular Nuclear Reactors For Commercial Shipping
 New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
New York Mandates Kill Switch and Surveillance Software in Your 3D Printer ...
 Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
Cameco Sees As Many As 20 AP1000 Nuclear Reactors On The Horizon
 His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
His grandparents had heart disease.
At 11, Laurent Simons decided he wanted to fight aging.
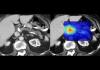 Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Mayo Clinic's AI Can Detect Pancreatic Cancer up to 3 Years Before Diagnosis–When Treatment...
Electrified cloth extracts uranium from seawater

By current estimates, there are about 8 million tonnes of known reserves of uranium on land. That's enough to fuel the world's nuclear reactors for centuries based on current technology, but in the sea there is an estimated 4.5 billion tonnes in the form of dissolved uranyl ions. If we could extract this economically, it would vastly extend our energy future. Even better, as uranium is removed from seawater, more would leach in from the Earth's crust, providing our descendants with over a billion years worth of nuclear fuel at any projected scale.
Led by Rui Zhao and Guangshan Zhu, the Northeast Normal team is looking at a novel way to extract these radioactive riches. Extraction isn't a new idea. In the past, other researchers have looked at using polymer mats, conductive fibers, and other methods. Now, Northeast Normal is looking at a flexible cloth woven from carbon fibers coated with two specialized monomers and treated with hydroxylamine hydrochloride. The porous cloth provides tiny pockets for the amidoxime, which captures the uranyl ions.
The capture itself seems almost like a school chemistry experiment in its simplicity. The cloth was placed in either seawater or a solution of uranyl ions where it acted as a cathode. Meanwhile, a graphite anode was added. When a current was run between the two, bright yellow, uranium-based precipitates accumulated on the cathode cloth in the same way that bronze coats a baby shoe as a parental memento.
In tests, the team reported extracting 12.6 mg of uranium per gram of water over 24 days, which is a higher amount and at a faster rate than other materials tested or simply allowing uranium to naturally accumulate on cloth.

 The Losertarian Problem
The Losertarian Problem


